There have been a number of reports of increased incidences of whooping cough (Bordetella pertussis (BP)) in the U.S., with clinical cases having risen throughout the last decades, primarily affecting teens and young adults, including those vaccinated when young. Having conducted further research to determine potential causes for the increase, scientists at La Jolla Institute for Immunology (LJI) have found that the shift in the 1990s from whole-cell pertussis (wP) vaccines to acellular pertussis (aP) vaccines may be a contributing factor. Over the next few years, the incidence of whooping cough rising in countries, such as the U.S. , England, Canada, Australia, Ireland, and Spain – all of which had switched to the new vaccine.
Authors of a F1000 Research article found that the second generation vaccine had an “unanticipated limitation” – that the vaccine prevents pertussis symptoms, but “has little impact on preventing people from becoming ‘colonized’ with the bacteria, meaning they are asymptomatic carriers of the disease and are still capable of infecting others.” The second generation vaccine was developed because the original vaccine contained so many bacterial components, it led to severe side effects in some.
The research points to the need for a new vaccine, one that does not lead to the severe side effects of the first, but prevents by symptoms and infection. As is seen in the annual reevaluation and development of the flu vaccine, and most recently the new COVID vaccine, diseases and/or our understanding of them continually evolve, creating the need for continually evolving vaccines.
With the continued evolution, TAG encourages people to stay up to date – on both the science and the current vaccine recommendations. We urge those 60 and older to get the RSV vaccine, as the virus can have severe repercussions, and follow CDC advice on who and when to get flu and COVID vaccines as well.
For more information on a vast array of infectious diseases, including whooping cough/pertussis, influenza, and COVID, visit TAG’s Public Health webpages to view and download any of our Infectious Disease Fact Sheets.
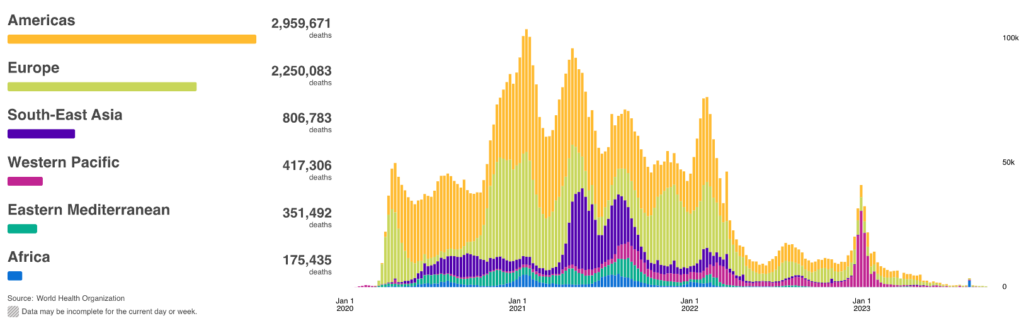
COVID Risk Matrix:
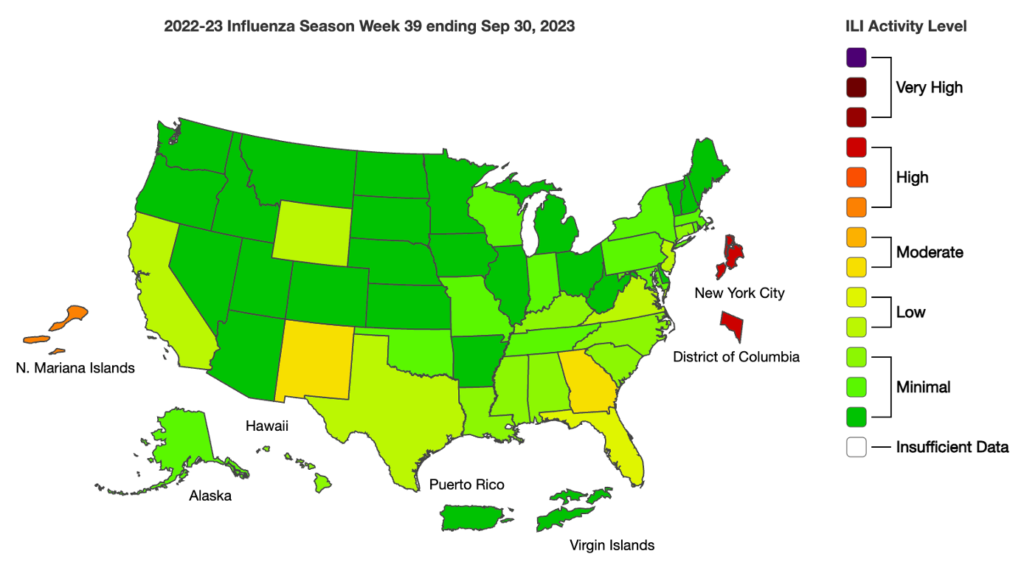
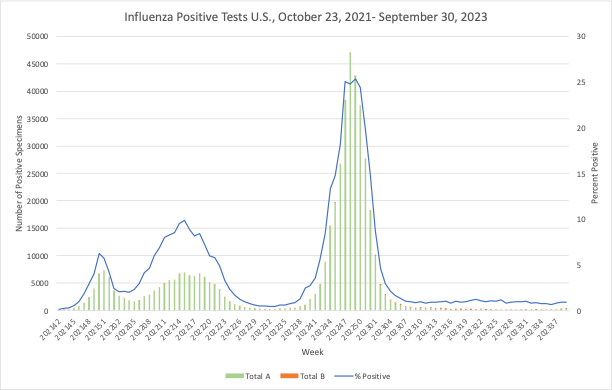
Influenza:
Infectious Disease News:
- During February 2022–May 2023, hospitalizations for RSV were less frequent but were associated with more severe disease than were hospitalizations for COVID-19 or influenza, including receipt of standard flow oxygen therapy, high-flow nasal cannula or noninvasive ventilation, and intensive care unit admission. Among hospitalized older adults, RSV was less common, but was associated with more severe disease than COVID-19 or influenza. The potential for severe RSV disease among older adults is important to consider as part of shared clinical decision-making when assessing the benefit of RSV vaccination among adults aged ≥60 years.
- The World Health Organization has recommended a new vaccine, R21/Matrix-M, for the prevention of malaria in children. The R21 vaccine is the second malaria vaccine recommended by WHO, following the RTS,S/AS01 vaccine, which received a WHO recommendation in 2021. Both vaccines are shown to be safe and effective in preventing malaria in children and, when implemented broadly, are expected to have high public health impact. Malaria, a mosquito-borne disease, places a particularly high burden on children in the African Region, where nearly half a million children die from the disease each year. Demand for malaria vaccines is unprecedented; however, available supply of RTS,S is limited. The addition of R21 to the list of WHO-recommended malaria vaccines is expected to result in sufficient vaccine supply to benefit all children living in areas where malaria is a public health risk.
- A press release from Arkansas Department of Health reported that there was a locally acquired malaria infection, which marks the fourth state this year to report a locally acquired case. The individual did not travel outside of the country.
- A study from Morbidity and Mortality Weekly Report identified that nearly 90% of all COVID-related deaths in 2023 in the US were among older adults – 65 years of age and older.
- Last week the FDA granted emergency use authorization for Novavax’s updated COVID vaccine. The new version includes the spike protein XBB.1.5 Omicron variant. The vaccine is available for individuals 12 years of age and older.