Key Points:
COVID
- In today’s Recommendation for Industry, we discuss TAG’s weekly updates on infectious disease and some further advice in reducing norovirus transmission. Read more below.
- Canada to make ArriveCan optional, drop vaccine requirement at the border by Sept. 30. The federal government plans to drop the COVID-19 vaccination requirement for people who enter Canada by the end of September, the same day it ends random testing of arrivals and makes optional the ArriveCan app. The changes, for arrivals at land, air and seaports of entry, are planned for Sept. 30, but have yet to be finalized by cabinet. The mask requirement for people on trains and planes will remain. Most European countries dropped entry requirements related to COVID-19 before summer began, returning to prepandemic standards in a bid to welcome tourists after more than two years of widespread illness, lockdowns and stay-home orders. The United States requires all entrants to be vaccinated, and it is not known if the requirement will be lifted at the same time as Canada’s.
- Global COVID-19 cases stabilize as deaths drop. After a 5-week drop, global COVID-19 cases stabilized last week, though deaths continued to decline, the World Health Organization (WHO) said in its weekly update on the pandemic. Of more than 3.2 million new cases reported last week, the five countries reporting the most were Japan, the United States, South Korea, Russia, and China. Meanwhile, deaths declined 17% from the previous week, with countries reporting more than 9,800 fatalities to the WHO. Amid reports of shortages of Moderna’s updated booster shots, the agency will soon release 10 million doses that were held back due to safety reviews at an Indiana plant that is bottling the packaging the product, according to the Washington Post.
Monkeypox
- Monkeypox Vaccines:
- Portugal approves preventive monkeypox vaccination for risk groups. Portugal has received 2,700 doses of the Bavarian Nordic’s (BAVA.CO) Imvanex vaccine in July and has started vaccinating close contacts. Portugal’s health authority widened its monkeypox vaccination strategy to include preventive shots for groups most at risk and approved the use of smaller doses, an approach known as “dose-sparing”, due to limited supplies.
- Hong Kong to start monkeypox vaccination on October 5. Hong Kong will launch a monkeypox vaccination programme for “high-risk groups” from Oct. 5 following the discovery of an imported case of monkeypox this month. Those groups include healthcare workers, laboratory personnel and people with “high-risk sexual practices” such as sex workers.
Food Safety & Public Health
- Uganda reports Ebola Sudan virus outbreak. Uganda’s health ministry today announced an Ebola Sudan virus outbreak in the central part of the country, with one confirmed case involving a man who died and eight suspected infections in people who are now receiving medical care. The man who passed away was confirmed to be a 24-year-old from Mubende district. In a statement, the World Health Organization (WHO) said the health ministry had been investigating six suspicious deaths in the district this month and that eight suspected patients are receiving medical care in a healthcare facility. Globally, there have been seven earlier Ebola Sudan outbreaks, four in Uganda and three in Sudan, with the most recent in Uganda in 2012.
- FDA leader updates infant formula status. For the past several months, the U.S. Food and Drug Administration has been working around the clock with our U.S. government partners, including the U.S. Department of Health and Human Services and the U.S. Department of Agriculture, to expand consumer access to infant formula products, while also ensuring that these products meet the agency’s safety, nutrition and quality standards. Yesterday, Dr. Solomon released the results of the review in a 10-page report, which includes information gleaned from interviews with FDA staff directly involved in the agency’s response to infant formula shortages as a result of the Sturgis facility closure. The report released yesterday highlights detailed findings and recommendations that will support the agency’s ongoing efforts to ensure that our most vulnerable population has consistent access to infant and specialty formula products in the future. Importantly, it also identifies the need for additional resources and authorities that will ensure the agency can fulfill our consumer protection role and gain important visibility into the supply chain with the aim of preventing these issues in the future.
- CDC issues Salmonella alert for people traveling to Mexico. Public health officials in the United States are warning travelers who have spent time in Mexico to be aware of multidrug-resistant strains of Salmonella Newport. The Centers for Disease Control and Prevention reports that some travelers have been infected with the strains, which have developed the ability to defeat drugs designed to kill them. “Many travelers with MDR (multidrug-resistant) Salmonella Newport infections reported eating beef, cheese — including queso fresco and Oaxaca— beef jerky, or dried beef — carne seca — before they got sick,” according to the alert from the CDC. The CDC has provided a list of recommendations for individuals who plan to travel to Mexico here.
- Risk Assessment of Norovirus Transmission in Food Establishments. This risk newly published assessment continues the analysis of the dynamics of norovirus transmission from ill or infected food employees in food establishments (restaurant setting). The previously published FDA quantitative risk assessment model in Duret et al. 2017, was used to evaluate more than 60 scenarios examining the impact of implementation and compliance with recommendations in the FDA Food Code for: restaurant surface cleaning and sanitizing, hand hygiene and employee health. For more information on FDA Food Code recommended intervention strategies for preventing norovirus, see Retail Food Protection.
Recommendations for Industry
Infectious Diseases on Positive Trend; Handwashing Remains Key Practice
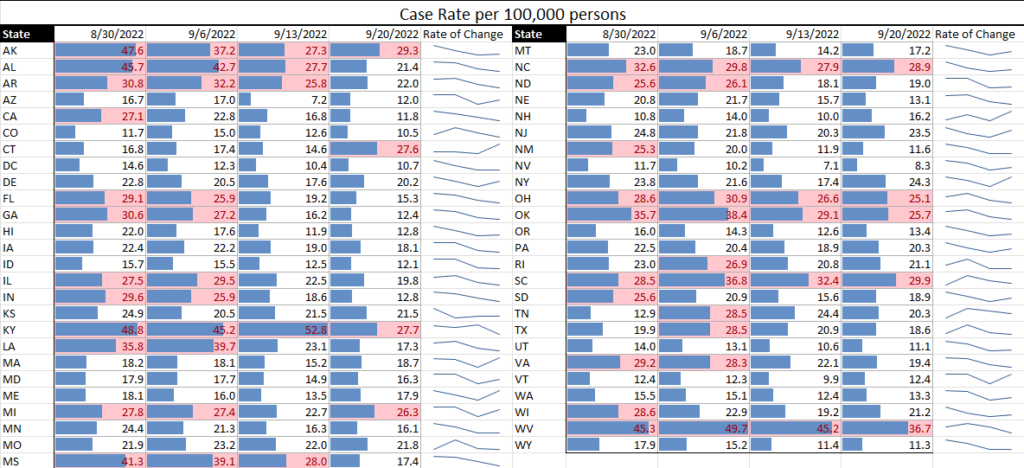
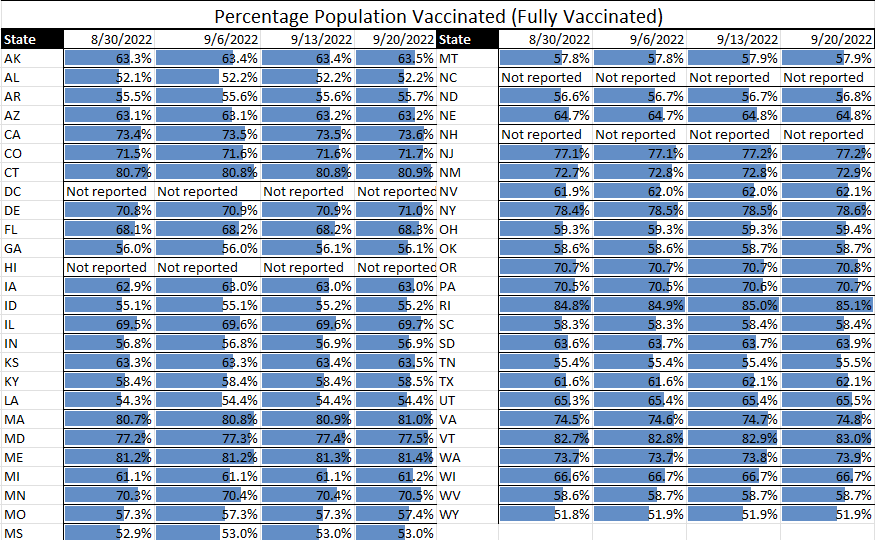
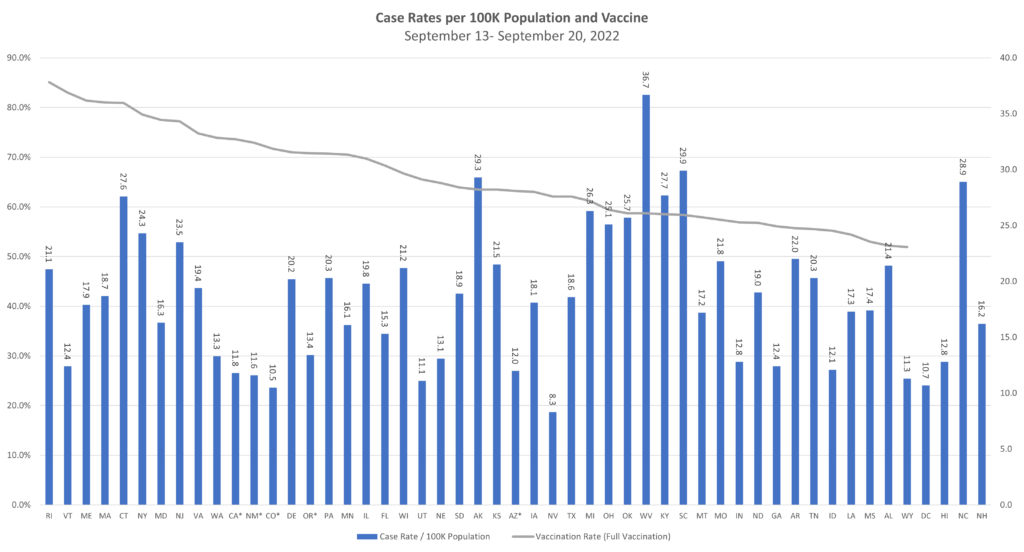
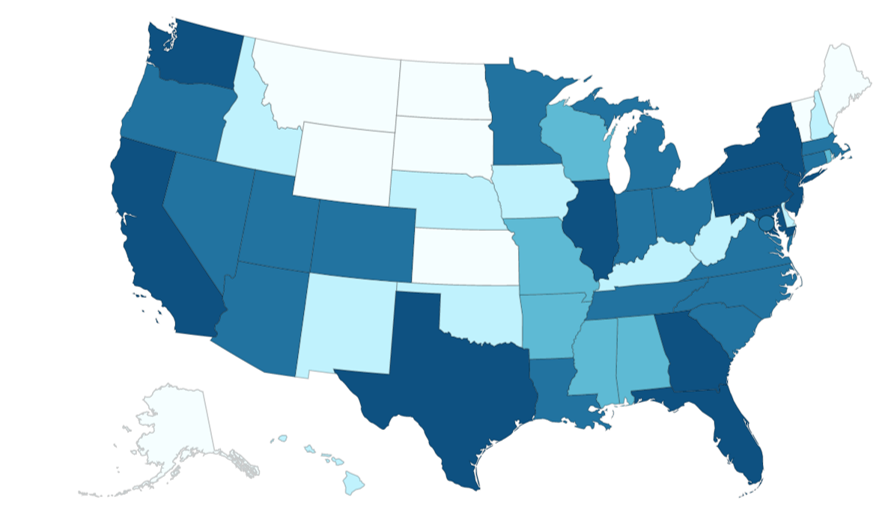
TAG’s Infectious Disease Update and Matrices (see graphs below) are showing all infectious diseases being tracked as being in positive states over the last week:
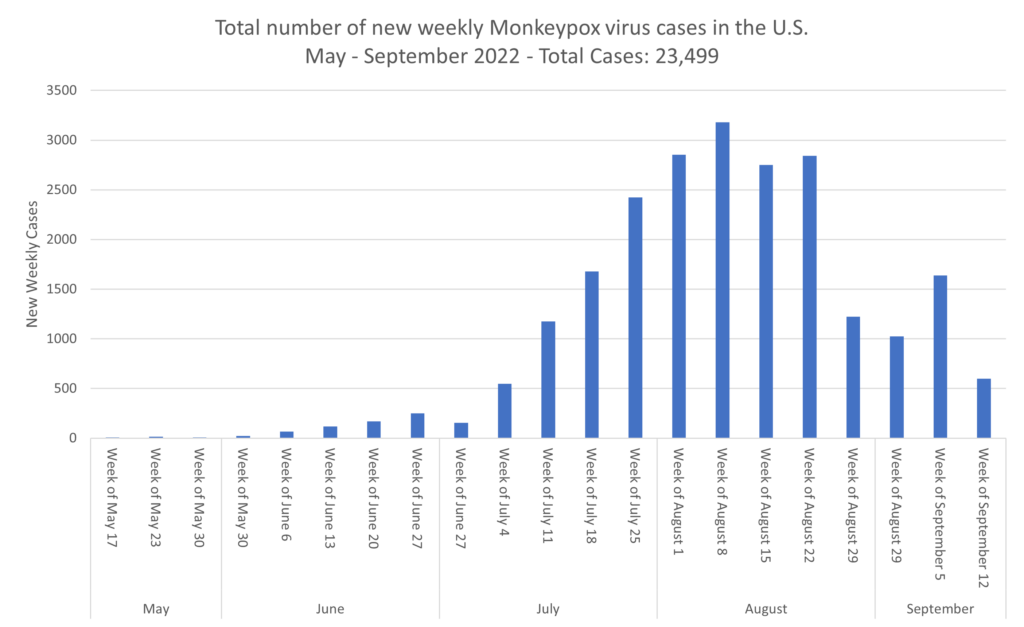
- Monkeypox is on a definite decline in the U.S.
- COVID continues to decline, with even hospital rates at fairly low levels
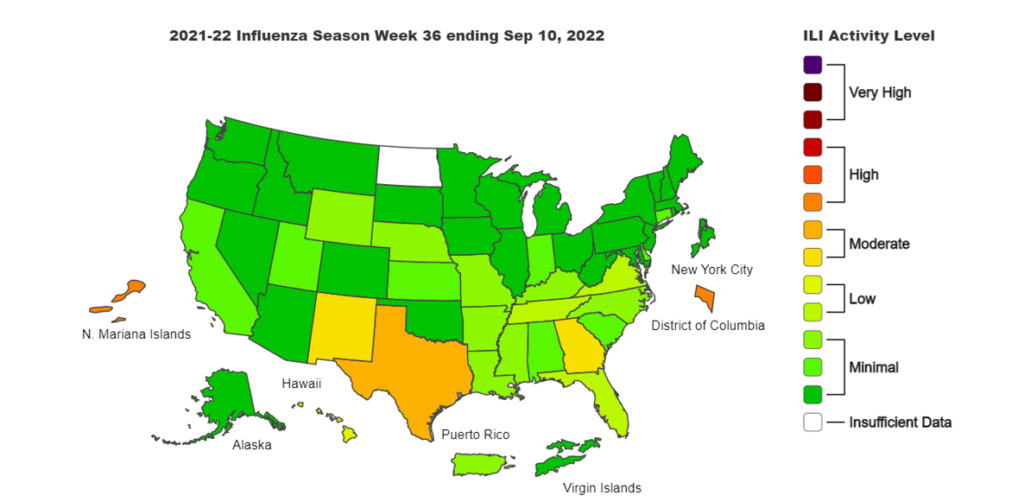
- Flu is beginning to tick up in some states, such as Texas, New Mexico, Georgia and the Southeast, but it remains low in most of the country.
We are seeing other respiratory illness on somewhat of a rise, but that is fairly typical of the changing seasons. We would also expect flu to begin trending upward in other areas, but we’re currently in a good situation there.
Norovirus. Earlier this week, we discussed the trending highs and lows of norovirus and recommendations for continuing to monitor worker health and implement protections. To expand on those recommendations, a study published recently by FDA risk assessors which focused on mitigation strategies for restaurants provides some targeted advice for all food establishments. Highlights of this work, which included modeled scenarios, found the following that can be considered for managing risks from norovirus:
- Compliance with excluding ill employees at the first sign of symptoms and hand hygiene rules had the most impact on consumer illnesses.
- Washing hands before donning and changing gloves efficiently reduces norovirus transfer. Proper handwashing has long been a key practice in maintaining food safety, and as this work shows, it continues to be critical in reducing transmission of both infectious and foodborne disease.
- Restriction of food employees (removed from contact with food and food-contact equipment and utensils) needs additional provisions to be effective.
- Surface cleaning and sanitizing has the least impact on consumer illnesses attributable to norovirus. Regardless it is still important to perform these practices correctly to minimize ither risks.
Risk Matrix:
Monkeypox:

Influenza:
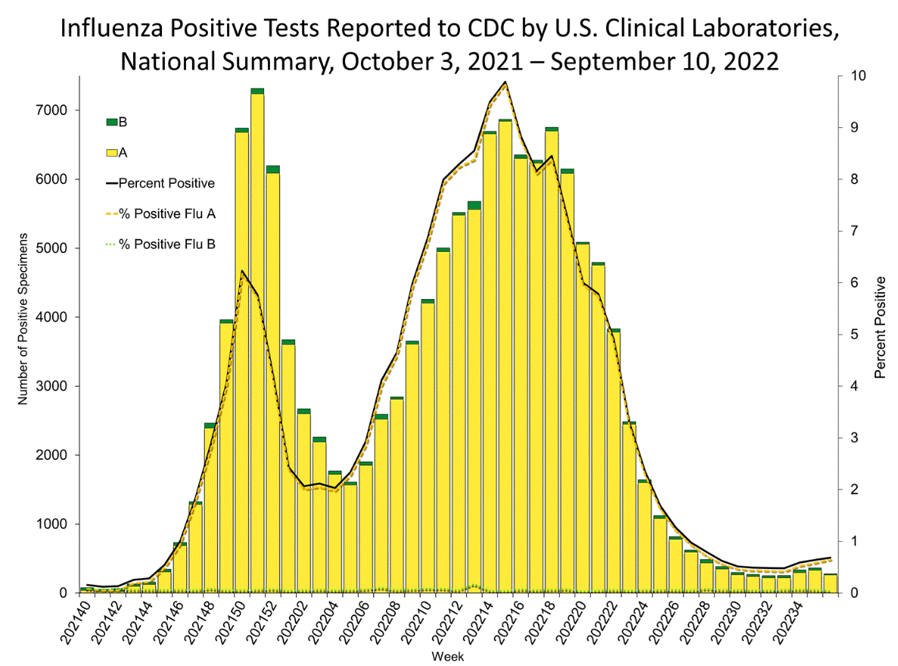
In case you missed it:
COVID
- In Tuesday’s Recommendation for Industry, we discussed norovirus cases. Read more here.
- Updated COVID booster tied to strong Omicron immune response. The new bivalent (two-strain) Moderna COVID-19 vaccine booster triggered stronger neutralizing antibody responses against the highly transmissible Omicron variant at 28 days than the previously authorized booster. The bivalent booster generated antibody geometric mean concentrations (GMCs) of 727.4 (95% CI, 632.8 to 836.1) and 492.1 (95% CI, 431.1 to 561.9) against the Omicron BA.4 and BA.5 subvariants, respectively (GMT ratio, 1.69; 95% CI, 1.51 to 1.90). The newer booster also produced greater binding antibody responses against previous strains such as Alpha, Beta, Gamma, and Delta (GMT ratios, 1.11 [95% CI, 1.03 to 1.19] to 1.24 [95% CI, 1.14 to 1.35]). Over median follow-ups of 43 days for the bivalent booster and 57 days for the monovalent version, rates of adverse events within 7 days of vaccination were similar, and most were mild or moderate. In both groups, the most common local reaction was pain at the injection site, while the most common systemic reactions were fatigue, headache, and muscle and joint pain. A total of 18.5% of the bivalent group and 20.7% of the monovalent group experienced adverse events 28 days or more after the second booster. Of those, 5.7% and 5.8%, respectively, were considered vaccine-related, and none were severe.
- Biden says ‘the pandemic is over’ even as death toll, costs mount. “We still have a problem with COVID. We’re still doing a lotta work on it. But the pandemic is over. If you notice, no one’s wearing masks. Everybody seems to be in pretty good shape. And so I think it’s changing,” Biden stated. Nearly 400 people a day continue to die from COVID-19 in the United States, according to the U.S. Centers for Disease Control and Prevention. Biden has asked Congress for $22.4 billion more in funding to prepare for a potential fall case surge.
- When should I get the Omicron booster? Will I need COVID shots forever? The Centers for Disease Control and Prevention is recommending that all teenagers and adults get an updated coronavirus booster shot designed to target both the original virus and the omicron variant circulating now. You’re eligible to get the updated booster shot if you’re 12 or older and it has been at least two months since your initial vaccine or your last booster. Older Americans, those with chronic illnesses and people who are immunocompromised or pregnant should get the updated booster shot as soon as possible. The CDC doesn’t expect side effects for the updated booster shot to differ from those associated with the current vaccines, which include redness and swelling where the shot was administered, as well as occasional fatigue, headache, and muscle soreness. The CDC says people who recently had a COVID-19 infection should consider waiting three months from their first symptoms, or positive test, before getting a booster shot. According to the CDC, young men who recently got the monkeypox vaccine, called Jynneos, might want to wait four weeks before getting any coronavirus vaccine. You can get the flu vaccine and a coronavirus vaccine or booster at the same time. But some experts said that to get maximum flu protection, you should get the coronavirus booster now and your flu shot in late October or early November. It is believed that there will be another booster in the fall of 2023 as well and that Americans will only need a booster once a year.
Monkeypox
- Florida infant, New Hampshire child contract monkeypox. Contracted from household surfaces and contacts, two children have been recently diagnosed with monkeypox, one from Florida and the other from New Hampshire. To date, 27 confirmed cases of monkeypox have been in pediatric aged patients. The CDC currently reports a nation total of 23,499 cases. In a preprint study, researchers in Singapore report isolating viable monkeypox virus from surfaces and dust samples, but they obtained no viable virus from air and water samples from a patient’s hospital room. A case report describes a possible tattoo-transmitted monkeypox infection in Italy. The patient reportedly got a tattoo in Spain in the 7 days prior to rash development. The rash developed where the tattoo needle was inserted.
- Low risk of monkeypox spread noted in health workers. In a report today of 313 healthcare workers (HCWs) exposed to monkeypox in Colorado, none of them contracted the virus, despite few wearing the recommended personal protective equipment (PPE) or receiving postexposure prophylaxis (PEP) vaccination. Most encounters between HCWs and monkeypox patients were short, with 69% lasting only 5 to 30 minutes. Only one HCW was exposed for more than 3 hours. Only seven (2%) were exposed during an aerosol-generating procedure. The CDC announced the launch of a new initiative called the Monkeypox Vaccine Equity Pilot Program. The program allows local, state, and territorial health departments, as well as tribal governments and non-governmental organizations, to submit requests to access the monkeypox vaccine, and it aims to reach populations that may face barriers to monkeypox vaccination. In related news, both New York and Oregon recently expanded access to monkeypox vaccines to include anyone at risk of exposure (in New York), and anyone who anticipates having or has had recent skin-to-skin contact and who knows someone who has had monkeypox, regardless of sexual preference or gender (Oregon). Also yesterday, the CDC updated treatment guidelines for the use of the antiviral Tpoxx (tecovirimat), recommending the drug be limited to people at high risk for severe disease. “Consider severe disease when a patient has conditions such as hemorrhagic disease; large number of lesions such that they are confluent; sepsis; encephalitis; ocular or periorbital infections; or other conditions requiring hospitalization,” the CDC said. Pregnant women and children over 8 years old should also be offered treatment.
Food Safety & Public Health
- Reported from Healthmap.org:
- Norovirus in the UK – cases of virus are 49 percent higher than what is expected of this time of year. Settings of cases are increasing in care homes and educational settings such as schools, colleges, and universities. Reports of measles have been recorded due to lack of vaccination in Zimbabwe, Paraguay, Afghanistan, and one case in Australia (linked to travel).
- One report stated that even if a child survives a case of measles, Harvard research shows that they may experience a phenomenon known as “immune amnesia” which wipes out existing immunity to other previous illnesses, leaving them even more susceptible to subsequent infections. This illustrates the need to work hard to combat this disease. The World Health Organization has activated its emergency response strategy in Zimbabwe to deal with the latest measles outbreak in the Southern African nation.
- Patient count grows in outbreak associated with Daily Harvest frozen crumbles. Federal officials continue to receive complaints of illnesses associated with frozen “Lentil & Leek Crumbles” sold by Daily Harvest. As of Sept. 15, the Food and Drug Administration had received 386 consumer complaints, up from 369 complaints reported on Aug. 25. Sick people have reported gastrointestinal illness and abnormal liver function, according to the FDA. Additionally, at least 25 patients had to have their gallbladders removed. Of known patients, 130 have required hospitalization. The company initiated a recall of its Lentil & Leek Crumbles on June 17.
- Three dead in Austrian Listeria outbreak. Käserei Gloggnitz has recalled a number of products in relation to the incident. Käserei Gloggnitz’s recall covers all kajmak, drinking yogurt and cream cheese products due to potential Listeria monocytogenes contamination. Kajmak is a type of cheese. Cluster analyses by the Austrian Agency for Health and Food Safety (AGES) found eight infections have occurred since 2020 due to an identical Listeria strain. Three people died between 2020 and 2022.





